
III. Description: Females
have a large serrated ovipositor,
unusual among the drosophilids. This atypical ovipositor is illustrated in
several fact sheets (Acheampong
2010, Walsh et al. 2010). In females of native
drosophilids, the teeth on the ovipositor are apparent, but
the structure appears less developed and overall
sclerotization is less
pronounced. In some images, the ovipositor is
shown exerted, but at rest it is
nestled beneath the tip of the abdomen. Males have a characteristic black spot near the ends of the wings. Several traits useful for identification of SWD, and several similar-looking
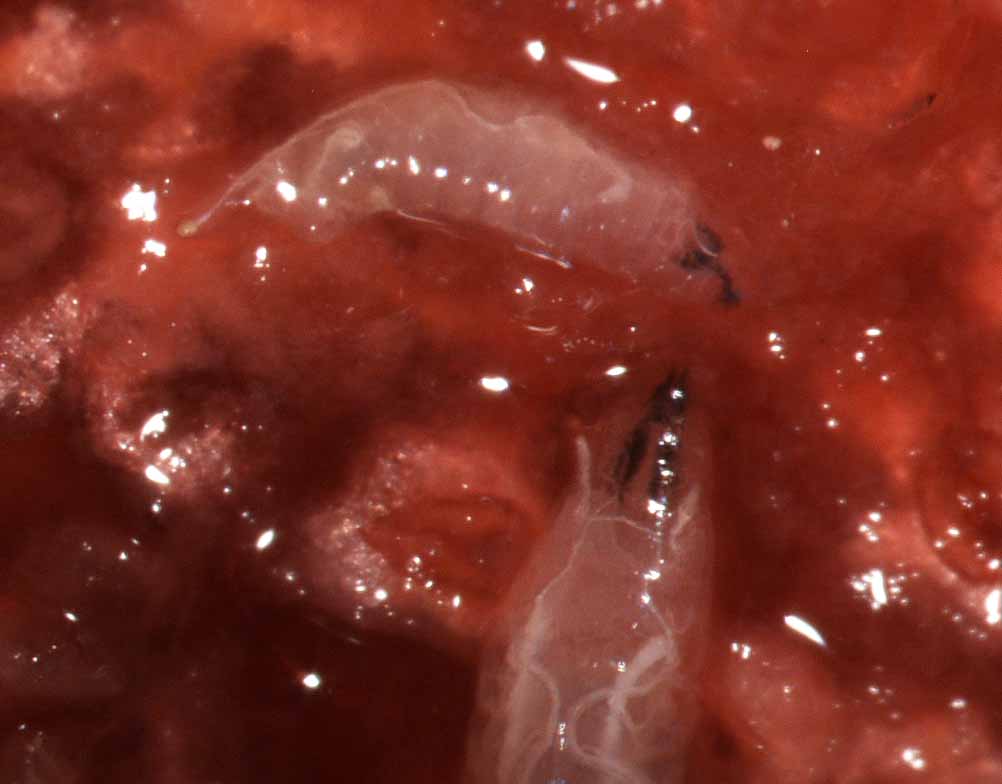
species, are shown in a factsheet from Michigan State University. Larvae
are translucent maggots 2-3 mm long, with black mouth hooks
visible at the anterior end. Silvery white tracheal
tubes may be visible through the dorsal cuticle with
magnification. Respiratory projections are present on
the posterior end.
Images of SWD life
stages:
Egg
Larva
Pupa
Adult female
Adult male
IV. Biology: In eastern Asia, there are up to 13 generations. A life cycle can be completed in 8-14 days, but adults can live up to 9 weeks. Females use the atypically large ovipositor to lay eggs in fruits as they are ripening, earlier than other drosophila species. Eggs are inserted under the skin of ripening fruit; each female lays 7-16 eggs/day. (See YouTube video showing oviposition behavior) Long respiratory horns project from one end of the egg. These horns may be found projecting from oviposition sites with magnification. Eggs hatch in 1-3 days, and larval feeding on the flesh causes a collapse of localized tissue after another 2 days, followed by growth of fungal or bacterial organisms; yeasts may be carried on the ovipositor (Walsh et al. 2010). Larvae are slender white maggots; pupae are brown, about 3 mm long, with two small respiratory horns protruding from one end. A circle of bristles surrounds the tip of the pupal respiratory projection.
V. Injury: Cherries were reported to have 70-80% injury by SWD, with eggs laid in sound fruit (Demerc (1965)). Crop losses of blueberries, caneberries and cherries have been reported ranging from
33-100% (Lehnert 2010). SWD has been found attacking wine grapes following veraison (see photo by Christine Vrooman). Berries may take on a shriveled, shrunken appearance, with small maggots feeding in the interior. The respiratory horns typical of drosophilid eggs may be seen in grapes as they approach harvest; see photos by Meredith Shrader of oviposition sites in Petit verdot and Viognier.
VI. Monitoring: In general, traps are
not powerful enough to serve as a control. Traps should be
used to detect activity, and when flies are detected, make
sure that other control measures are in place. Several commercial
traps are available (Trece and Scentry). A trapping
guide has been posted (Wallingford et al. 2018), with discussion of several baits,
and comparing commercial with homemade traps. Traps
using homemade baits of either yeast or apple cider vinegar
may be used for monitoring. Traps using apple cider
vinegar (ACV) alone are attractive to flies and less odorous
to work with than with yeast added; traps with added yeast
may be somewhat more sensitive, but fluid should be replaced
with each service of the traps. A combination of
ACV and red wine (60:40) is more attractive than ACV alone (Shrader 2017). If available, brown rice vinegar is more attractive than ACV (Willbrand and Pfeiffer 2019). Traps should be checked at least weekly. Most of the Drosophila
flies collected will not be SWD, so the flies collected must be checked carefully.
Control:
Chemical control
Control measures are directed against the adults; there are no effective controls for larvae in the fruit.
As vulnerable fruit approach ripeness, weekly spray applications should be made. Because of the high number of offspring and number of generations, there is a high risk of development of insecticide resistance. Consequently, insecticides with different mode
of actions should be rotated to prolong the effective life of
insecticides. Organophosphates (malathion and phosmet)
are effective (check labels for registrations on specific
crops), as are pyrethroids (be wary of induction of secondary
pests). Spinosyns offer an additional mode of action
class, with spinetoram having greater efficacy than
spinosad. See this Table for a list of insecticides available for SWD on the most
vulnerable crops. Included are the maximum number of
applications (or amounts of material) allowed per season and
the IRAC class of mode of action. This is important in
designing rotations - it will be helpful to reserve materials
effective against SWD until properly timed for that
pest. Organically approved treatments are included in
the table, but see the accompanying article for a fuller
discussion of organic approaches.
Our Southern Region Small Fruit Consortium
provides recommendations for SWD in the pest management guides
for caneberries, blueberries, strawberries and bunch grapes. In addition,
individual states may maintain small fruit management guides
that are helpful. In addition to the Pest Management Guides, the consortium
publishes a quarterly Small Fruit News. A recent issue contained articles
on conventional and organic management of SWD.
See this table for available SWD materials with preharvest, mode of
action, and maximum allowed applications. Organically
approved treatments are included in the links provided; the number
of organic tools is limited however, and management in organic
berries will be a challenge.
The following
links may be used for chemical control recommendations:
Virginia Cooperative Extension
Southeast Regional Small Fruit Consortium
Cultural control:
Netting of 80g weight was effective in controlling injury by SWD
(McDermott and Nickerson 2014,
Leach et al. 2016, Riggs et al. 2016, Ebbenga et al. 2019).
Lighter grades
(larger mesh) are not effective.
While netting is initially expensive, it becomes cost
effective because it may be used for several years.
Harvest fruit promptly and thoroughly to eliminate
breeding sites. It is important to harvest all fruit, including
those in the interior and lower parts of the plant canopy. This can be
problematic in pick-your-own operations. This issue should be
kept in mind once SWD established in an area, since at times
grape growers may leave berries on the vine to allow greater
development of some harvest parameters. Any overripe
or rotten fruit nearby should be destroyed. In vineyards,
pomace produced during the crushing process should not be dumped
near the producing vineyard block. This can become a
source for many SWD.
When berries are harvested, it will be helpful to
get them as cool as possible, as soon as possible. There is complete
mortality of larvae in fruit held for 96 hours at 35˚F, and
below 40˚F, eggs and larvae don’t develop (Bolda 2010,
Burrack 2016)
. In
most cases, such uniform holding conditions are not
maintained; fruit cooling should be considered a component of
SWD management and not a sole control tactic.
Biological
Control: Because of the ability of SWD to encapsulate and kill the eggs of our native parasitoid wasps, biological control has not been successful. Research is underway to find parasitic species that are able to attack this species.
VIII.
Larvae in your berries or cherries?
With head capsule:
With legs: Caterpillar - cherry fruit
worm, cranberry fruitworm, or oriental fruit moth
Lacking legs: Plum curculio
Lacking head capsule:
With respiratory projection on hind end,
tapering at both ends, 2-3 mm long: Spotted wing drosophila
Lacking respiratory horns, tapering at front
end, broad at rear end, 5-6 mm long: Cherry fruit fly, blueberry maggot
IX. An additional exotic drosophilid!
In September 2012, an additional exotic drosophilid was found to
be common in some vineyard blocks. The African fig fly, Zaprionus indianus is originally from Africa but in recent years has been expanding its range.
From Brazil it moved northward, and was found in Florida in 2005
and South Carolina in 2007. When present, concurrent
infestation with SWD generally existed. However in one
block, Zaprionus was far more common than SWD. AFF
may follow SWD by laying eggs in or near ovipostion punctures
created by SWD. This fly is red-brown in color, with
longitudinal white body stripes, thinly bordered by black.
See adult flies in dorsal
and oblique view.
This fly is also described in a recorded presentation linked here.
Acheampong, S. 2010. Spotted wing drosophila (Drosophila suzukii), a new vinegar fly pest in British Columbia. Brit. Columbia Minist. Agric. Lands, Kelowna BC Bolda, M. P., R. E. Goodhue and F. G. Zalom. 2009. Spotted wing drosophila: Potential economic impact of a newly established pest. Giannini Fndn. of Agric. Economics, Univ. of Calfornia.
Burrack, H. 2016. Preventing and managing spotted wing drosophila infestation. NC State Extension. Entomology - Insect Biology and Management. https://entomology.ces.ncsu.edu/2016/06/preventing-and-managing-spotted-wing-drosophila-infestation/
Ebbenga, D. N., E. C. Burkness, and W. D. Hutchison. 2019. Evaluation of exclusion netting for spotted-wing drosophila (Diptera: Drosophilidae) management in Minnesota wine grapes. J. Econ. Entomol. 112: 2287–2294.
Fisher, P., H. Fraser, D. Beaton and L. Huffman. 2012. Spotted Wing Drosophila: A New Threat To Tender Fruit And Berry Crops. Ontario Minist. Agriculture, Food and Rural Affairs.
Leach, H., S. Van
Timmeren, and R. Isaacs. 2016. Exclusion netting
delays and reduces Drosophila suzukii (Diptera: Drosophilidae) infestation in
raspberries. J. Econ. Entomol. 109: 2151-2158.
Lehnert, D. 2010. New fruit fly is more than just a nuisance. Fruit Growers News 49: 12.
McDermott, L., and L.
Nickerson. 2014. Evaluation of insect exclusion and
mass trapping as cultural controls of spotted wing
drosophila in organic blueberry production. N. Y. Fruit
Quarterly 11: 25-27.
Milkovich, M. 2010. SWD found in Michigan; team planning a response, pp. 1,14, Fruit Growers News, Sparta MI.
Pfeiffer, D. G. 2012. Caneberry pest management: Borers, thrips and Virginia State Univ., Petersburg. Mar 15. Recorded presentation.
Pfeiffer, D. G., T. C. Leskey and H. J. Burrack. 2012. Threatening the harvest: The threat from three invasive insects in late season vineyards. p. 449-474. In: N. J. Bostanian, C. Vincent and R. Isaacs (eds.). Arthropod Management in Vineyards: Pests, Approaches, and Future Directions. Springer, Dordrecht, The Netherlands. 505 p.
Wallingford, A., B.
Sideman, and G. Hamilton. 2018. Monitoring spotted
wing drosophila (SWD) with traps. Univ. New Hampshire
Extension. https://extension.unh.edu/resource/monitoring-spotted-wing-drosophila-swd-traps.